- Visibility 86 Views
- Downloads 18 Downloads
- DOI 10.18231/j.ijor.2023.006
-
CrossMark
- Citation
Human anterior cruciate ligament-derived mesenchymal stem cells for regenerative medicine applications
Background
The anterior cruciate ligament (ACL) is the most commonly injured ligament in the knee[1] at a rate of 1 in 3000 individuals in the United States annually.[2], [3], [4] The ACL exhibits a poor self-healing ability attributed partly to its intrinsic nature (impediment to cell migration) and partly to the environmental niche in which it lies (synovial fluid, poor blood supply, and clot formation).[2], [5], [6], [7], [8], [9], [10] Although some authors have resurrected arthroscopic repair techniques, [11] reconstructive surgery utilizing autografts and/or allograft have traditionally been utilized to treat ACL injuries, with around 100,000 procedures performed annually within the United States. [1], [3], [4], [5], [6], [7], [8], [9], [12], [13], [14], [15] However, graft-based approaches have limitations including graft rejection, disease transmission, lack of adequate blood supply, donor site morbidity, and limited availability. [3], [4], [14], [16], [17], [18], [19], [20], [21], [22], [23]
Recently, the use of mesenchymal stem cells (MSCs)in this field has been proposed [2], [5], [11], [14], [15], [24], [25], [26], [16], [27]. MSCs can differentiate towards adipogenic, osteogenic and chondrogenic lineages, have fibroblast like morphology, are able to self-renew, and give rise to tissues/organs such as brain, muscle, liver, and kidney. [2], [5], [10], [28], [29], [30], [31], [32], [33], [34], [35] MSCs have been obtained from sources such as bone marrow and adipose tissue, existing virtually in all tissues within the perivascular niches. They have also been successfully isolated from various tissues such as trabecular bone, teeth, skin, muscle, synovial fluid, peripheral blood, heart, lungs, spleen, kidney, intestine, trachea, prostate, nasal mucosa, umbilical cord blood, placenta, amniotic fluid, and Wharton’s jelly. [2], [14], [25], [28], [29], [30], [36], [37], [38], [39] MSCs are expandable, immunosuppressive and do not stimulate an immediate immune response: they are thus popular candidates for regenerative medicine and cell therapy applications.[40], [41] MSCs have the potential to regenerate the ACL, thanks to their ability to self-renew, differentiate and secrete cytokines, growth factors and extracellular matrix components including collagen [5], [42]. MSCs are also reported to have the ability to regenerate torn ACL, meniscus and cartilage following intraarticular administration [2]. Although both BMSCs and ACL derived MSCs could be induced to ligament specific fibroblasts, the latter expressed higher levels of mRNAs for collagen types I and III. [14]
Cellular therapies utilizing MSCs are based on isolation of unselected autologous cells from the bone marrow or isolation of specific sub-populations. [40], [41], [42], [43], [44] One of the most essential properties for isolation and purification of MSCs is plastic adherence. [45] Several techniques have been used for isolation and enrichment of MSCs such as antibody-based cell sorting, density-based culture techniques, positive and negative selection methods.[46], [47], [48], [49], [50], [51] Irrespective of the isolation methodology and the heterogenous nature of the MSCs, isolated MSCs should satisfy specific criteria. In 2006, the International Society for Cellular Therapy (ISCT) Mesenchymal and Tissue Stem Cell Committee proposed the specific criteria to define human MSCs that include: plastic adherence; positive for CD73/5’-Nucleotidase, CD90/Thy1, andCD105/Endoglin; negative for hematopoietic markers including CD34, CD45, CD11b, CD14, CD79 alpha or CD19 and HLA- DR surface molecules; and the ability to differentiate to osteoblasts and adipocytes in vitro. [30], [34], [36], [38], [40], [52], [53], [54]
Currently, there is limited literature demonstrating the isolation of MSCs from ACL as well as a lack of adequate isolation methodology with proper adherence to ISCT criteria. [2], [14], [55] Thus, the goal of this study was to isolate and characterize MSCs from ACL adhering to the aforementioned ISCT criteria. We believe that the experimental variability and contradiction in data using various different methods to isolate, culture, differentiate, and define mesenchymal stem cells (MSCs) can be minimized when adhering to the criteria defined by the International Society for Cellular Therapy (ISCT) in 2006. Based on these criteria, we hypothesize that we can harvest MSCs from hACL that can be used for reconstruction of ACL. To test this hypothesis, we will isolate MSCs from hACL based on markers defined by ISCT in 2006; validate the isolated cells are in fact MSCs by various differentiation and clonogenic studies; and characterize the isolated MSCs in terms of their phenotypic characteristics.
Materials and Methods
Isolation and expansion of MSCs
Two discarded ACL tissue samples were procured from patients undergoing total knee reconstruction at the Memorial Medical Center (Springfield, IL, USA). The patients’ age, sex and name were kept anonymous. Since the tissue was a medical waste, exemption of approval was granted by the Institutional Review Board at Southern Illinois University School of Medicine (Springfield, IL, USA). The samples were transported to the laboratory in refrigerated normal saline. Upon reaching the laboratory, the tissues were minced and digested with 0.1% (w/v) Collagenase-I (Sigma-Aldrich, St. Louis, MO, USA) for 16h in α-MEM medium containing 10% FBS and 1% Pen-Strep overnight at 370C under humid condition with 5% CO2. After incubation, cells were centrifuged at 1500rpm for 5min and suspended in the same medium. Cells were then recovered via 70µm nylon mesh cell strainer (MIDSCI, St. Louis, MO, USA) and washed twice with the same medium. The cells isolated from FACS were plated in α-MEM medium containing 10% FBS and 1% Pen-Strep overnight at 370C under humid condition with 5% CO2. When approaching confluency, cell morphology was studied at light microscopy.
Immuno profiling
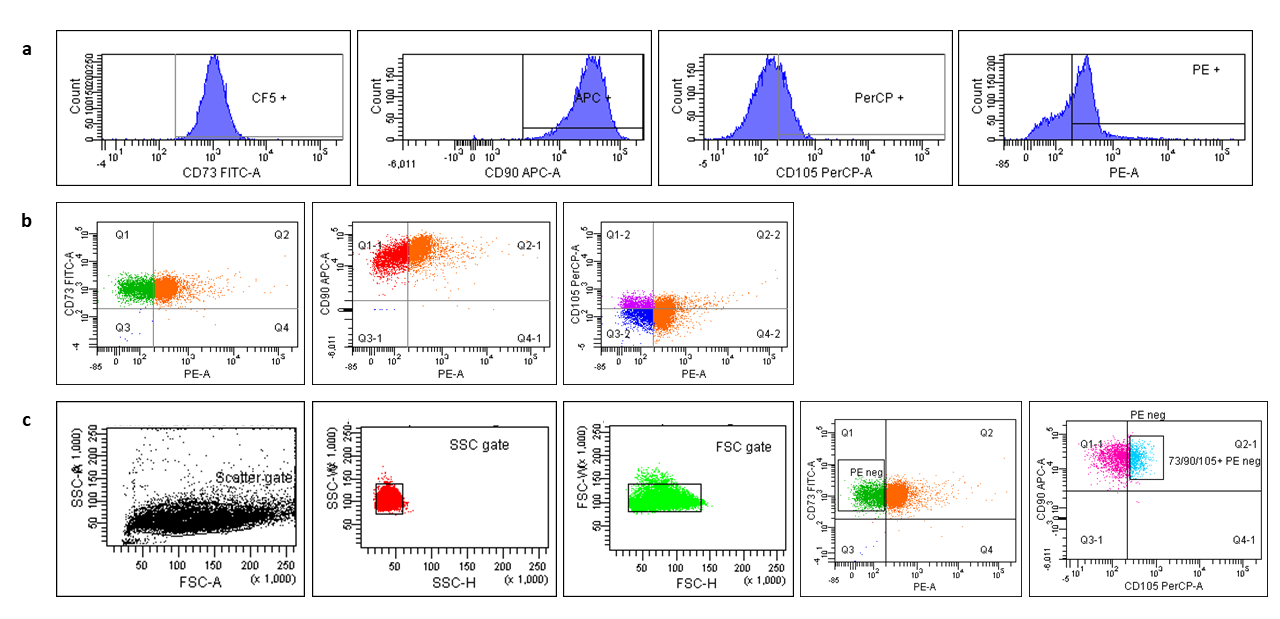
The cells were then prepared for fluorescence activated cell sorting (FACS) analysis based on the standard antibodies used for identification of MSCs as defined by the ISCT following manufacturer instructions (R&D systems, Minneapolis, MN, USA; Cat. FMC020). Briefly, about 4ml of cell suspension was pelleted by centrifugation at 1500rpm for 5min and suspended in 2ml of staining buffer from the kit. The test tube was then centrifuged at 300g for 5min, decanted and suspended in 1ml of the aforementioned buffer. The cells were then counted by hemocytometry and diluted to the density of 105cells /100µl of staining buffer in a series of seven tubes, each containing a) CD90-APC (10µl) b) CD73-CFS (10µl) c) CD105-PerCP (10µl) d) Negative (PE) marker cocktail (10µl) (Hematopoietic markers including CD34, CD45, CD11b and CD14; CD79 alpha and CD19 alpha; and HLA Class II) e) all three positive Isotype control (10µl) f) CD90 (10µl) + CD73 (10µl) + CD105 (10µl) + Negative marker cocktail (µl) and a tube containing only 105cells /100 µl of staining buffer was used as a background control. The tubes were then incubated for 30-45min at room temperature (RT) in the dark. After incubation, excess antibodies were washed by adding 2ml of staining buffer and centrifuging at 1500rpm for 5min. The cells were then suspended in 400µl of staining buffer and transported to the core facility for FACS by using Becton-Dickinson FACSAriaII high-speed cell sorter (Franklin Lakes, NJ, USA).
Immunocytochemistry
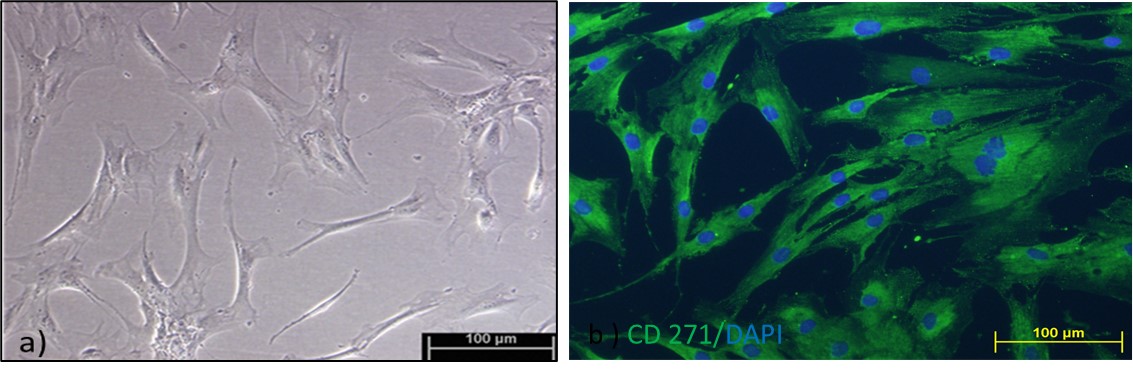
P-3 cells were grown in three wells of a 48 well plate (Corning Inc., Corning, NY, USA) and confirmed for the presence of MSCs using universal CD271marker. For this, the cells were washed twice with 1ml phosphate buffer saline (PBS) and fixed with 0.2ml of 4% paraformaldehyde in PBS for 20min at RT. The cells were then washed three times with 0.2ml of 1% bovine serum albumin (BSA) in PBS for 5min followed by subsequent permeabilization and blocking with 0.2ml of 0.3% Triton X-100, 1% BSA and 10% normal donkey serum in PBS at RT for 45min. After blocking, the cells were incubated with 100µl/well of mouse Mab CD271(R & D Systems, Minneapolis, MN, USA; Cat. No: MAB367) prepared in PBS containing 0.3% Triton X-100, 1% BSA and 10% normal donkey serum to a final concentration of 20µg /ml overnight at 2-80C. A negative control was run using PBS containing 0.03% Triton X-100, 1% BSA and 10% normal donkey serum with no primary antibody. The cells were washed three times with 0.2ml of 1% BSA in PBS for 5min followed by incubation with 100-300µl/well secondary goat anti-mouse Alexa Fluor 488 (Cell Signaling, Danvers, MA, USA; Cat.no. 4408S) antibody prepared in 1% BSA in PBS at 1: 1000 dilution, and incubated in dark for 60min at RT. The cells were then washed three times with 0.2ml of 1% BSA in PBS for 5min. Afterwards, incubation with nuclear stain DAPI (Molecular probes, Life technologies, Carlsbad, CA, USA) for 2minutes was performed followed by washing with PBS for three times, 5min each. The cells were then covered with 0.5ml of PBS and visualized under fluorescence microscopy.
Population characteristics
Growth curve
P-2 cells plated in all wells of a 12 well plate at the seeding density of 5 × 103cells/well were allowed to become confluent. The medium was changed every third day. Three wells were then trypsinized every third day and counted. The growth curve was then plotted using the counting data.
Over agar assay
A modification of soft agar assay known as “over agar” assay was used to verify anchorage independent growth of the MSCs [2]. Briefly,10000 P-2cells in 2ml of complete growth medium were plated on top of pre-hardened 1% agarose (0.5 ml) in three wells of a six well plate. The plates were then incubated at 370C in a humidified atmosphere of 5% CO2 and 95% air for 14 days. The medium was changed every third day. Colonies were verified using an inverted phase contrast microscope.
Colony forming assay
For colony forming assays, about 100 P-2 cells were plated in triplicates in 100mm tissue culture dishes (Corning Inc., Corning, NY, USA) in complete culture medium. Cells were maintained in complete medium for 14d at 370C in 5% humidified CO2, with media changes every fourth day. On day 14, the cells were washed with PBS and fixed in 4% formaldehyde for 15min at RT, followed by incubation for 5min at RT in 0.5% Trypan blue solution in ethanol. The plates were then washed twice with distilled water, dried, and the resulting colony forming units were counted under light microscopy.
Population doubling (PD)
Three wells of a six well plate were seeded with 10000 P-2cells/well and grown to confluence with medium changes every third day. The cells were then trypsinized and counted every fifth day. Subsequent sub-culture was performed at the same seeding density of 10000 cells/well in triplicate. Passaging was performed to the sixth passage (P-6). First, the population doubling number (PDN) was determined with the following formula: PDN =log (N/N0) x 3.31; where PDN = the population doubling number, N = cell number at the end of the culture period (five days), N0= the initial number of cells (1.5 x 105 cells); second, the population doubling time (PDT) was calculated with the help of formula: PDT = CT/PDN; where PDT = population doubling time, CT = the duration of culture (5 days).
Lineage differentiation assays
Adipogenic, Osteogenic and Chondrogenic Potential
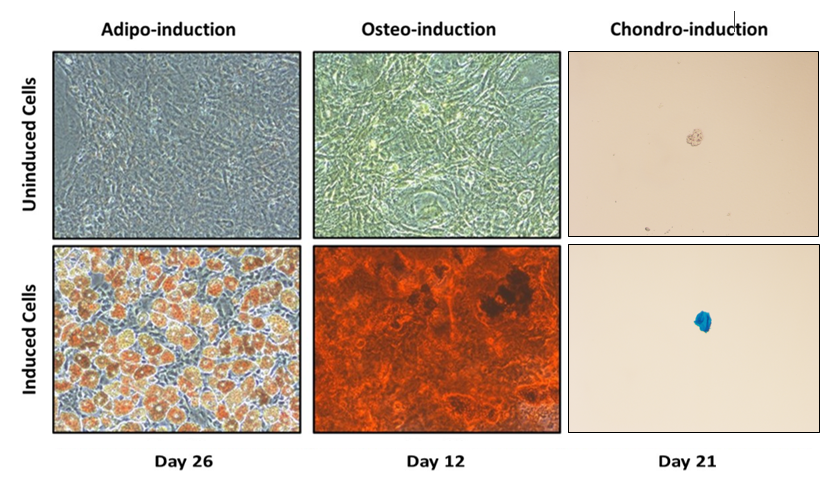
The multipotency of the isolated cells was determined by inducing the cells towards osteoblast, adipocyte and chondrocyte lineages. P3-cells in 6200cells/well for osteoblast lineage, and 42,000cells/well for chondrocyte and adipocyte lineage were seeded in 24-well plates. Cells were allowed to grow overnight for the osteoblasts or until cells were confluent for chondrocytes and adipocytes (approximately 3-5 days with media changes every 2 days) in an incubator at 37⁰C with 5% CO2 overnight. When the cells were ready for induction, plates were divided into 2 groups. Group 1 continued to receive the regular culture medium and group 2 received the osteo-, chondro- and adipo-induction medium (Lonza, USA). The medium was changed according to the manufacturer’s instructions in both groups until the cells showed signs of induction. Alizarin Red (Sigma-Aldrich, St. Louis, MO), Oil Red O (Sigma-Aldrich, St. Louis, MO) and Alcian Blue (Sigma-Aldrich, St. Louis, MO) stains were used according to manufacturer’s instructions to confirm osteo-, adipo- and chondro-inductions, respectively.
Cell cycle analysis
FACS tubes each containing 1x105h bone marrow- derived MSCs (BMSCs) at P-11 or 1x105ACL-MSCs at P-3 in triplicates were placed on ice prior to fixation. The cells were then washed with cold PBS and then suspended in 1ml of 80% ice-cold ethanol while pipetting up and down. The tubes were then incubated for 30min and washed twice with cold PBS. After incubation, the cells were suspended in 1ml of DAPI (1 µg/ml)/TX-100(0.1%) solution and incubated at RT for another 30min. Finally, cells were analyzed employing FACS by using Becton-Dickinson FACS Aria II high-speed cell sorter. The excitation used was 355nm whereas the emission was measured using a filter 450/50 nm.
Statistical analysis
A total of two biological replicates and three technical replicates were used in all experiments. One-way ANOVA (Analysis of Variance) and Student’s paired t-test were used for the comparison of means. p< 0.05 was considered significant. The analysis was performed using GraphPad Prism (version 6.01).
Results
Phenotypic profile of isolated MSCs
The results from the FACS analysis showed that the isolated cells were MSCs with the following immunoprofiling. The cells stained positive (≥95%) for i) CD73; CD90; CD105 and negative (≤2%) for following Hematopoietic markers: i) CD34, CD45, CD11b and CD14; CD79 alpha and CD19 alpha and HLA Class II. The average rate of isolation of ACL-MSC following our protocol was 5.5% (i.e., these cells expressed MSCs phenotypic markers) ([Figure 1]).
Phenotypic validation of FACS isolated MSCs
The isolated cells showed spindle shaped morphology under light microscopy ([Figure 2]a). Further immunocytochemistry results of these cells stained with CD271 cell surface marker indicates the potential MSC origin of the isolated cells ([Figure 2]b).
Population characteristics
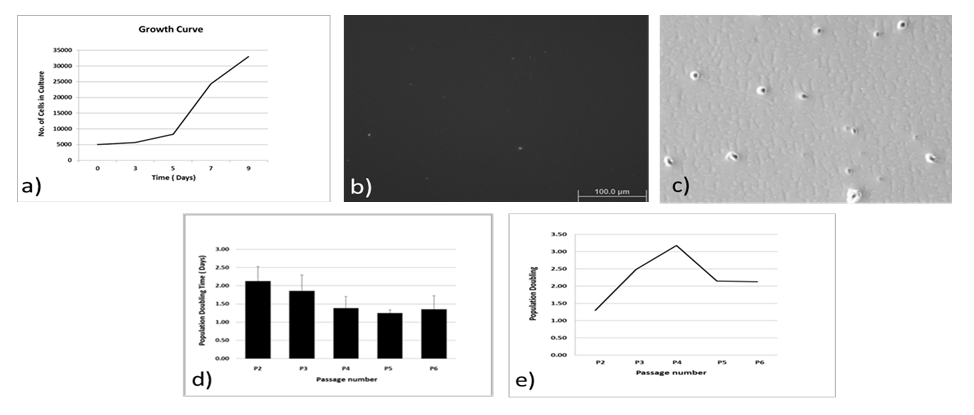
The growth behavior of the P3-MSCs which was followed for up to a period of 9 days showed a normal lag phase and a log phase. The over agar assay which is based on the principle to detect the anchorage independent growth of the cells failed to reveal growth of any distinct colony. However, some colonies could be seen on the plastic Petri dishes during the colony forming assay demonstrating the clonogenic potential of these isolated MSCs. PD study showed that the cells were rapidly dividing during the P-4 passage with PDT value of ~1.5 day, although the PDT values during the various passages were not statistically significant (One way ANOVA, p= 0.294) ([Figure 3]).
Adipogenic, Osteogenic and Chondrogenic potential
The multipotency of the isolated cells was demonstrated by their successful induction towards osteo-, adipo- and chondro- lineages. Alizarin Red, Oil Red O and Alcian Blue staining images demonstrated successful induction towards Osteo-, Adipo- and Chondro- lineages at Day 26, 12 and 21 respectively ([Figure 4]).
Cell cycle analysis
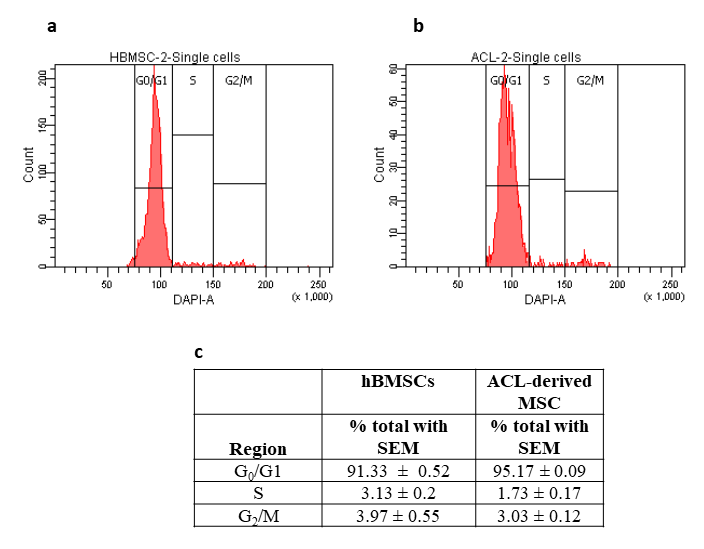
Results of the cell cycle analysis showed that there were no pre-apoptotic cells and significantly higher percentage of cells were found in G0/G1 for ACL-derived MSCs (Student’s paired t-test, p=0.0245) compared to hBMSCs. On the other hand, for the S phase, the reverse held true (Student’s paired t-test, p= 0.0067). For the G2/M phase, no statistically significant difference was found in the percentage of cells between the two groups (Student’s paired t-test, p=0.2911) ([Figure 5]).
Discussion
The ACL is one of the most commonly injured ligaments of the knee. [55], [56] An injured ACL has a poor prognosis, as it fails to regenerate itself from lack of adequate blood supply and continuous contact with synovial fluid. [3], [5], [57], [58] Thus, despite some advocates of direct end to end repair, ACL reconstruction is one of the few options, unless cell therapy is considered [3], [57]. MSCs have intrinsic properties to self-renew, differentiate into various lineages, and proliferate indefinitely, and their use for tissue based regeneration and repair seems promising [56]. In fact, they have been used successfully in the regeneration of bone, cartilage, and cardiovascular and neural tissues [2], [5], [25], [38], [59], [60].MSCs can be isolated from two main sources: adult tissues such as bone marrow, adipose tissue, peripheral blood; and fetal tissues such as placenta, umbilical cord, umbilical cord blood, amniotic fluid. [61] MSCs which are found within the adult organs are the remnants of prenatal cells which originally derived from the mesodermal layer of the embryo giving rise to bone, cartilage, tendon, fat, muscle and marrow stroma.[60] The tissue protective, reparative and regenerative abilities of MSCs are attributed mainly to their paracrine secretion of several trophic factors including, cytokines, chemokines and growth factors, whose actions can be realized locally or far from the actual site of injection.[25], [30], [43], [58] The mechanisms by which these factors act include targeting neighboring cells to produce functionally active mediators or directly altering the intracellular signaling in the injured tissues. This paves the way for the use of MSCs for cellular therapy by injecting them locally or systemically. In fact, local injection of adipose derived stem cells in the knee joint of mice showed promising results with inhibition of synovial activation and formation of chondrophyte/osteophyte together with inhibition of cartilage destruction possibly by suppressing synovial macrophage activation.[29], [59], [60] In addition, MSCs possess immunomodulatory properties from modifications in the secretome profile of the immune cells such as dendritic cells, natural killer cells and effector T cells and suppression of CD4+/CD8+ T–cell proliferation. [25], [16], [30], [34], [38], [52], [53] Furthermore, given the low expression of MHC-I, and the lack of MHC-II as well as costimulatory molecules (CD40, CD40L, CD80 and CD86), MSCs do not cause significant T-cell proliferation and essentially escape an immune response themselves. [61], [62], [63]
Most of what is known of MSCs arises from studies using BMSCs. BMSCs which were the first MSCs to be isolated, expanded and differentiated, have traditionally been used to compare to other MSCs from different lineages. Tissue specific MSCs all possess the characteristic immunomodulatory and regenerative capabilities of BMSCs, as well as being more geared towards differentiating towards their lineage [2], [14], [29], [33], [34]. Although both BMSCs and ACL-derived MSCs could be induced to ligament specific fibroblasts, the latter expressed higher levels of mRNAs for Collagen types I and III. [14] A recent study demonstrated that BMSCs differentiated better into bone whereas synovium-derived MSCs (Syn-MSCs) differentiated better into adipogenic and chondrogenic lineages, emphasizing the notion that it is better to use MSCs from the target tissue which needs repairing. [32] Given the high rate of proliferation and collagen production, researchers have started to consider MSCs for repair of the ACL.[53] MSCs can accelerate and improve tendon healing both in vivo and in vitro[64] by directly differentiating into tendon cells and secreting cytokines to modulate inflammation and tissue healing.[65] Yu et al. have shown that the exosome secreted by bone marrow MSCs promote tendon repair by proliferating and recruiting of endogenous tendon stem cells. [66], [67], [68]
MSCs isolated based on plastic adherence possess a mixture of heterogeneous populations differing both in phenotypes and functions, thus stressing the need to isolate MSCs based on ISCT criteria [37], [52]. Although the current criteria for isolation of MSCs help to isolate a well-defined population of cells from a heterogeneous mixture of cells, the definition lacks discriminatory power to discern MSCs from fibroblasts.[53] MSCs are relatively rare in the bone marrow, with a prevalence of 0.001-0.01% of nucleated cells. [37] In the present study, we were able to expand the number of cells with MSCs markers to 5.5% using an original enrichment technique. We acknowledge that may still be a mixture of stem cells and fibroblasts, but our subsequent experiments with MSCs specific marker (please see below) and differentiation into different lineages showed that at least the majority of the isolates were MSCs in nature. Flores-Torales et al. identified CD271 as a key marker that could define MSCs.[69] Our results are in accordance with this report and indicate the presence of CD271 in our isolated cells, confirmed via immunocytochemistry ([Figure 2]b). The cells also showed spindle shaped fibroblast morphology at light microscopy ([Figure 2]a). The growth curve of the isolated MSCs showed distinct lag phase and log phase. However, we could not see a stationary phase during the culture period of 9 days ([Figure 3]a). The over agar assay, a modification of soft agar assay, is used to detect malignant transformation of cells since cells which can grow in an anchorage independent manner (such as tumor cells)are only able to form colonies. [33], [37], [38], [70] In our experiments, the isolated MSCs did not form any significant colonies suggesting they were not transformed ([Figure 3]b). However, the isolated cells were able to form colonies on plastic culture dish, implicating their clonogenic potential, a characteristic feature of stem cells (Figure 3c). In this study, the PDTs in general for P2-P6 cells were approximately at or below 2 days ([Figure 3]d,e). Umbilical cord-derived MSCs (UC-MSCs) up to sixth passage (P6) had the PDT of 24h, in contrast, BMSCs had PDT of 40h for same passage number.[30], [71], [72] Similarly, PDTs of 45.2h or adipose derived MSCs (ASCs) and 61.2h for BMSCs have also been reported [30]. Our differentiation studies also showed that the MSCs were able to differentiate into adipocytes shown by positive Oil Red O stain ([Figure 4]). The isolated MSCs in our study were also able to differentiate into osteoblasts as demonstrated by positive Alizarin Red S staining ([Figure 4]), indicative of Ca++ mineralization.[16] For chondrogenic differentiation, cells were grown under induction medium: these cells were capable of producing proteoglycan rich extracellular matrices as revealed by positive Alcian blue staining ([Figure 4]). The analysis of cell cycle pattern ([Figure 5]) between the two cell lines, namely hBMSCs (P11) and ACL-derived MSCs (P3), suggested that the former cells were more proliferative than the latter since we used exactly the same number of cells for each case (105 cells). Therefore, it seems that ACL-MSCs spend most of their time in G0/G1 phase, unwilling to proliferate, giving rise to significantly higher number of cells at this phase of cell cycle.
Despite some promising preliminary data, our study is not without limitations. The number of samples evaluated was small. The BMSCs used were present at a later passage (P11) and might have lost their phenotypic characteristics. Furthermore, although the ACL samples were collected under ice, some possibility of tissue degradation could have occurred in the meantime, which we will attempt to avoid in the future studies. The immunocytochemistry analysis lacks analysis of other stem cell markers and negative control for CD271 marker. In the present study we did not isolate highly purified and single stem cells. In future studies, we intend to perform immunocytochemistry for other stem cell markers including CD271 negative control and attempt to isolate purified single stem cells. In addition, kinetic/mechanistic experiments along with in vivo studies to assess regenerative potential such as angiogenesis and immune tolerance are warranted to obtain robust confirmation regarding the reparative potential of the isolated cells.
Conclusion
In summary, the preliminary results from our experiments demonstrate that MSCs as defined by the ISCT criteria do exist in the ACL tissue. In addition, as ACLs are normally discarded during total knee reconstruction, collection of this discarded tissue to isolate MSCs offers less ethical concerns . With those reasons in mind along with studies suggesting that MSCs from a tissue specific lineage can lead to better differentiation into that target tissue, ACL-MSCs can be a potentially powerful source of MSC for future ACL reconstruction and regeneration when injected in the torn ligament of the knee after suitable expansion in-vitro. Given the small number of biological replicates used in this study, a future study involving larger sample size is warranted.
Key Messages
MSCs are present in human ACL and fulfil the criteria for stemness of the ISCT.
Tissue specific lineage may lead to better differentiation into that target tissue. Thus, ACL-derived MSCs can be a powerful source for future ACL reconstruction and regeneration.
Abbreviations
ANOVA-Analysis of Variance, BMSCs-Bone marrow-derived Mesenchymal Stem Cells, BSA-Bovine serum albumin, FACS-Fluorescence Activated Cell Sorting, ACL-Anterior Cruciate Ligament, ISCT-International Society for Cellular Therapy, MSCs-Mesenchymal Stem Cells, PBS-Phosphate buffer saline, PDT-Population Doubling Time, RT-Room temperature.
Availability of data and materials
Raw data were generated at Southern Illinois University School of Medicine, Springfield, Illinois, USA. Derived data supporting the findings of this study are available from the corresponding author [SFE] on request.
Author Contributions
RK and SFE conceptualized and designed the study. RK and MHR performed the experiments. RK, AG, NM, CA, MHR, and SFE analyzed the data and interpreted the results. RK and AG wrote the manuscript draft. RK, AG, NM, CA, MHR and SFE edited the manuscript. AG and SFE supervised the study. All authors have read and approved the final manuscript.
Ethics Approval and Consent to Participate
Ethical review and approval were waived by the Institutional Review Board at Southern Illinois University School of Medicine for this study, due to tissue collected being classified as medical waste and no protected health information was collected.
Source of Funding
This study was funded by Southern Illinois University School of Medicine, Springfield, Illinois, USA and the APC was funded by El-Amin Orthopaedic & Sports Medicine Institute, Lawrenceville, Georgia, USA.
Conflict of Interest
None.
Acknowledgements
The authors would also like to thank Dr. Darwin Prockop at Tulane Center, Tulane University for providing hBMSCs.
References
- DT Nguyen, TH Ramwadhdoebe, CP Van Der Hart, L Blankevoort, PP Tak, CN Van Dijk. Intrinsic healing response of the human anterior cruciate ligament: an histological study of reattached ACL remnants. J Orthop Res 2014. [Google Scholar]
- MT Cheng, HW Yang, TH Chen, OK Lee. Isolation and characterization of multipotent stem cells from human cruciate ligaments. Cell Prolif 2009. [Google Scholar]
- AM Kiapour, MM Murray. Basic science of anterior cruciate ligament injury and repair. Bone Joint Res 2014. [Google Scholar]
- A Gupta, ASR Potty, D Ganta, RJ Mistovich, S Penna, C Cady. Streamlining the KOOS Activities of Daily Living Subscale Using Machine Learning. Orthop J Sports Med 2020. [Google Scholar] [Crossref]
- D Figueroa, M Espinosa, R Calvo, M Scheu, A Vaisman, M Gallegos. Anterior cruciate ligament regeneration using mesenchymal stem cells and collagen type I scaffold in a rabbit model. Knee Surg Sports Traumatol Arthrosc 2014. [Google Scholar]
- BC Fleming, EM Magarian, SL Harrison, DJ Paller, MM Murray. Collagen scaffold supplementation does not improve the functional properties of the repaired anterior cruciate ligament. J Orthop Res 2010. [Google Scholar]
- F Bi, Z Shi, A Liu, P Guo, S Yan. Anterior cruciate ligament reconstruction in a rabbit model using silk-collagen scaffold and comparison with autograft. PLoS One 2015. [Google Scholar] [Crossref]
- J Zhang, T Pan, H J Im, F H Fu, J H Wang. Differential properties of human ACL and MCL stem cells may be responsible for their differential healing capacity. BMC Med 2011. [Google Scholar] [Crossref]
- Y Mifune, T Matsumoto, S Ota, M Nishimori, A Usas, S Kopf. Therapeutic potential of anterior cruciate ligament-derived stem cells for anterior cruciate ligament reconstruction. Cell Transplant 2012. [Google Scholar]
- T Matsumoto, S M Ingham, Y Mifune, A Osawa, A Logar, A Usas. Isolation and characterization of human anterior cruciate ligament-derived vascular stem cells. Stem Cells Dev 2012. [Google Scholar]
- R Papalia, G Torre, G Papalia, S Campi, N Maffulli, V Denaro. Arthroscopic primary repair of the anterior cruciate ligament in adults: a systematic review. Br Med Bull 2019. [Google Scholar]
- KP Spindler, MM Murray, JL Carey, D Zurakowski, BC Fleming. The use of platelets to affect functional healing of an anterior cruciate ligament (ACL) autograft in a caprine ACL reconstruction model. J Orthop Res 2009. [Google Scholar]
- T Zantop, W Petersen, F H Fu. Anatomy of the anterior cruciate ligament. Oper Tech Orthop 2005. [Google Scholar]
- TF Huang, YT Chen, TH Yang, LL Chen, SH Chiou, TH Tsai. Isolation and characterization of mesenchymal stromal cells from human anterior cruciate ligament. Cytotherapy 2008. [Google Scholar]
- A Gupta, K Sharif, M Walters, MD Woods, A Potty, BJ Main. Surgical retrieval, isolation and in vitro expansion of human anterior cruciate ligament - derived cells for tissue engineering applications. J Vis Exp 2014. [Google Scholar] [Crossref]
- L Trombi, L Mattii, S Pacini, H Patil, V Kumaraswamy, S Jha. The role of stem cells in osteoarthritis: An experimental study in rabbits. Bone Joint Res 2014. [Google Scholar] [Crossref]
- G I Im, NH Jung, SK Tae. Chondrogenic differentiation of mesenchymal stem cells isolated from patients in late adulthood: the optimal conditions of growth factors. Tissue Eng 2006. [Google Scholar]
- A Gupta, M D Woods, K D Illingworth, R Niemeier, I Schafer, C Cady. Single walled carbon nanotube composites for bone tissue engineering. J Orthop Res 2013. [Google Scholar]
- A Gupta, B J Main, B L Taylor, M Gupta, CA Whitworth, C Cady. In vitro evaluation of three-dimensional single-walled carbon nanotube composites for bone tissue engineering. J Biomed Mater Res A 2014. [Google Scholar]
- A Gupta, TA Liberati, SJ Verhulst, BJ Main, MH Roberts, AG Potty. Biocompatibility of single-walled carbon nanotube composites for bone regeneration. Bone Joint Res 2015. [Google Scholar]
- A Gupta, N Kukkar, K Sharif, BJ Main, CE Albers, SF El-Amin III. Bone graft substitutes for spine fusion: A brief review. World J Orthop 2015. [Google Scholar]
- A Gupta, SF El-Amin, HJ Levy, R Sze-Tu, SE Ibim, N Maffulli. Umbilical cord-derived Wharton's jelly for regenerative medicine applications. J Orthop Surg Res 2020. [Google Scholar] [Crossref]
- AGR Potty, A Gupta, H C Rodriguez, I W Stone, N Maffulli. Intraosseous Bioplasty for a Subchondral Cyst in the Lateral Condyle of Femur. J Clin Med 2020. [Google Scholar] [Crossref]
- I K Dhammi, H Rehan Ul, S Kumar, . Graft choices for anterior cruciate ligament reconstruction. Indian J Orthop 2015. [Google Scholar]
- PD Counsel, D Bates, R Boyd, DA Connell. Cell therapy in joint disorders. Sports Health 2015. [Google Scholar]
- A Singh, SC Goel, KK Gupta, M Kumar, GR Arun, H Patil. The role of stem cells in osteoarthritis: An experimental study in rabbits. Bone Joint Res 2014. [Google Scholar]
- A Gupta, C Cady, A M Fauser, H C Rodriguez, R J Mistovich, Agr Potty. Cell-free Stem Cell-Derived Extract Formulation for Regenerative Medicine Applications. Int J Mol Sci 2020. [Google Scholar]
- A Lotfy, M Salama, F Zahran, E Jones, A Badawy, M Sobh. Characterization of mesenchymal stem cells derived from rat bone marrow and adipose tissue: a comparative study. Int J Stem Cells 2014. [Google Scholar]
- M Maumus, C Jorgensen, D Noël. Mesenchymal stem cells in regenerative medicine applied to rheumatic diseases: role of secretome and exosomes. Biochimie 2013. [Google Scholar]
- R Hass, C Kasper, S Böhm, R Jacobs. Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Commun Signal 2011. [Google Scholar] [Crossref]
- K Neumann, T Dehne, M Endres, C Erggelet, C Kaps, J Ringe. Chondrogenic differentiation capacity of human mesenchymal progenitor cells derived from subchondral cortico-spongious bone. J Orthop Res 2008. [Google Scholar]
- Y Ogata, Y Mabuchi, M Yoshida, EG Suto, N Suzuki, T Muneta. Purified Human Synovium Mesenchymal Stem Cells as a Good Resource for Cartilage Regeneration. PLoS One 2015. [Google Scholar] [Crossref]
- V Sabapathy, B Sundaram, VM Sreelakshmi, P Mankuzhy, S Kumar. Human Wharton's Jelly Mesenchymal Stem Cells plasticity augments scar-free skin wound healing with hair growth. PLoS One 2014. [Google Scholar] [Crossref]
- JS Chen, VW Wong, GC Gurtner. Therapeutic potential of bone marrow-derived mesenchymal stem cells for cutaneous wound healing. Front Immunol 2012. [Google Scholar] [Crossref]
- A Navani, L Manchikanti, SL Albers, RE Latchaw, J Sanapati, AD Kaye. Responsible, Safe, and Effective Use of Biologics in the Management of Low Back Pain: American Society of Interventional Pain Physicians (ASIPP) Guidelines. Pain Physician 2019. [Google Scholar]
- T Tawonsawatruk, A Spadaccino, I R Murray, B Peault, H A Simpson. Growth kinetics of rat mesenchymal stem cells from 3 potential sources: bone marrow, periosteum and adipose tissue. J Med Assoc Thai 2012. [Google Scholar]
- N Sessarego, A Parodi, M Podestà, F Benvenuto, M Mogni, V Raviolo. Multipotent mesenchymal stromal cells from amniotic fluid: solid perspectives for clinical application. Haematologica 2008. [Google Scholar]
- WR Otto, NA Wright. Mesenchymal stem cells: from experiment to clinic. Fibrogenesis Tissue Repair 2011. [Google Scholar] [Crossref]
- B J Main, JA Valk, N Maffulli, HC Rodriguez, M Gupta, IW Stone. Umbilical cord-derived Wharton's jelly for regenerative medicine applications in orthopedic surgery: a systematic review protocol. J Orthop Surg Res 2020. [Google Scholar] [Crossref]
- K Baghaei, SM Hashemi, S Tokhanbigli, A Asadi Rad, H Assadzadeh-Aghdaei, A Sharifian, . Isolation, differentiation, and characterization of mesenchymal stem cells from human bone marrow. Gastroenterol Hepatol Bed Bench 2017. [Google Scholar]
- A Gupta, S Kashte, M Gupta, HC Rodriguez, SS Gautam, S Kadam. Mesenchymal stem cells and exosome therapy for COVID-19: current status and future perspective. Hum Cell 2020. [Google Scholar]
- D Kouroupis, A Kyrkou, E Triantafyllidi, M Katsimpoulas, G Chalepakis, A Groussia. Generation of stem cell-based bioartificial anterior cruciate ligament (ACL) grafts for effective ACL rupture repair. Stem Cell Res 2016. [Google Scholar]
- AI Caplan, D Correa. The MSC: an injury drugstore. Cell Stem Cell 2011. [Google Scholar]
- D Kouroupis, C Sanjurjo-Rodriguez, E Jones, D Correa. Mesenchymal Stem Cell Functionalization for Enhanced Therapeutic Applications. Tissue Eng Part B Rev 2019. [Google Scholar]
- T Tondreau, L Lagneaux, M Dejeneffe, A Delforge, M Massy, C Mortier. Isolation of BM mesenchymal stem cells by plastic adhesion or negative selection: phenotype, proliferation kinetics and differentiation potential. Cytotherapy 2004. [Google Scholar]
- R F Suijkerbuijk, DE Olde Weghuis, M Van Den Berg, F Pedeutour, A Forus, O Myklebost. Comparative genomic hybridization as a tool to define two distinct chromosome 12-derived amplification units in well-differentiated liposarcomas. Genes Chromosomes Cancer 1994. [Google Scholar]
- MB Eslaminejad, S Nadri. Murine mesenchymal stem cell isolated and expanded in low- and high-density culture system: surface antigen expression and osteogenic culture mineralization. In Vitro Cell Dev Biol Anim 2009. [Google Scholar]
- MB Eslaminejad, A Nikmahzar, L Taghiyar, S Nadri, M Massumi. Murine mesenchymal stem cells isolated by low density primary culture system. Dev Growth Differ 2006. [Google Scholar]
- M Baddoo, K Hill, R Wilkinson, D Gaupp, C Hughes, GC Kopen. Characterization of mesenchymal stem cells isolated from murine bone marrow by negative selection. J Cell Biochem 2003. [Google Scholar]
- M Soleimani, S Nadri. A protocol for isolation and culture of mesenchymal stem cells from mouse bone marrow. Nat Protoc 2009. [Google Scholar]
- V A Siclari, J Zhu, K Akiyama, F Liu, X Zhang, A Chandra. Mesenchymal progenitors residing close to the bone surface are functionally distinct from those in the central bone marrow. Bone 2013. [Google Scholar]
- J A Ankrum, J F Ong, J M Karp. Mesenchymal stem cells: immune evasive, not immune privileged. Nat Biotechnol 2014. [Google Scholar]
- G Kundrotas. Surface markers distinguishing mesenchymal stem cells from fibroblasts. Acta Medica Lituanica 2012. [Google Scholar] [Crossref]
- H C Rodriguez, M Gupta, E Cavazos-Escobar, S F El-Amin, A Gupta. Umbilical cord: an allogenic tissue for potential treatment of COVID-19. Hum Cell 2021. [Google Scholar]
- A Gupta, K Sharif, M Walters, M D Woods, A Potty, B J Main. Surgical retrieval, isolation and in vitro expansion of human anterior cruciate ligament-derived cells for tissue engineering applications. J Vis Exp 2014. [Google Scholar] [Crossref]
- DH Lee, J Ng, SB Kim, CH Sonn, KM Lee, SB Han. Effect of Donor Age on the Proportion of Mesenchymal Stem Cells Derived from Anterior Cruciate Ligaments. PLoS One 2015. [Google Scholar] [Crossref]
- MM Murray, BC Fleming. Biology of anterior cruciate ligament injury and repair: Kappa delta ann doner vaughn award paper 2013. J Orthop Res 2013. [Google Scholar]
- R Mardones, CM Jofré, JJ Minguell. Cell Therapy and Tissue Engineering Approaches for Cartilage Repair and/or Regeneration. Int J Stem Cells 2015. [Google Scholar]
- D Kouroupis, A C Bowles, M Williman, C P Orfei, A Colombini, T M Best. Infrapatellar fat pad-derived MSC response to inflammation and fibrosis induces an immunomodulatory phenotype involving CD10-mediated Substance P degradation. Sci Rep 2019. [Google Scholar] [Crossref]
- D Kouroupis, A Bowles, T M Best, L D Kaplan, D Correa. CD10/Neprilysin Enrichment in Infrapatellar Fat Pad-Derived Mesenchymal Stem Cells Under Regulatory-Compliant Conditions: Implications for Efficient Synovitis and Fat Pad Fibrosis Reversal. Am J Sports Med 2020. [Google Scholar]
- A Kanaya, M Deie, N Adachi, M Nishimori, S Yanada, M Ochi. Intra-articular injection of mesenchymal stromal cells in partially torn anterior cruciate ligaments in a rat model. Arthroscopy 2007. [Google Scholar]
- CJ Centeno, SJ Faulkner. The Use of Mesenchymal Stem Cells in Orthopedics. Stem Cells Cancer Stem Cells 2012. [Google Scholar]
- Z Mohammadi, JT Afshari, MR Keramati, DH Alamdari, M Ganjibakhsh, AM Zarmehri. Differentiation of adipocytes and osteocytes from human adipose and placental mesenchymal stem cells. Iran J Basic Med Sci 2015. [Google Scholar]
- R Costa-Almeida, I Calejo, M E Gomes. Mesenchymal Stem Cells Empowering Tendon Regenerative Therapies. Int J Mol Sci 2019. [Google Scholar] [Crossref]
- F Migliorini, M Tingart, N Maffulli. Progress with stem cell therapies for tendon tissue regeneration. Expert Opin Biol Ther 2020. [Google Scholar]
- H Yu, J Cheng, W Shi, B Ren, F Zhao, Y Shi. Bone marrow mesenchymal stem cell-derived exosomes promote tendon regeneration by facilitation the proliferation and migration of endogenous tendon stem/progenitor cells. Acta Biomater 2020. [Google Scholar] [Crossref]
- M J Martínez-Lorenzo, M Royo-Cañas, E Alegre-Aguarón, P Desportes, T Castiella, F García-Alvarez. Phenotype and chondrogenic differentiation of mesenchymal cells from adipose tissue of different species. J Orthop Res 2009. [Google Scholar]
- E Flores-Torales, A Orozco-Barocio, O R Gonzalez-Ramella, A Carrasco-Yalan, K Gazarian, S Cuneo-Pareto. The CD271 expression could be alone for establisher phenotypic marker in Bone Marrow derived mesenchymal stem cells. Folia Histochem Cytobiol 2010. [Google Scholar]
- A S Dighe, S Yang, V Madhu, G Balian, Q Cui. Interferon gamma and T cells inhibit osteogenesis induced by allogeneic mesenchymal stromal cells. J Orthop Res 2013. [Google Scholar]
- G V Røsland, A Svendsen, A Torsvik, E Sobala, E Mccormack, H Immervoll. Long-term cultures of bone marrow-derived human mesenchymal stem cells frequently undergo spontaneous malignant transformation. Cancer Res 2009. [Google Scholar]
- A Gupta, N Maffulli, HC Rodriguez, CE Lee, HJ Levy, F El-Amin. Umbilical cord-derived Wharton's jelly for treatment of knee osteoarthritis: study protocol for a non-randomized, open-label, multi-center trial. J Orthop Surg Res 2021. [Google Scholar] [Crossref]
- A Gupta, N Maffulli, HC Rodriguez, EW Carson, RA Bascharon, K Delfino. Safety and efficacy of umbilical cord-derived Wharton’s jelly compared to hyaluronic acid and saline for knee osteoarthritis: study protocol for a randomized, controlled, single-blind, multi-center trial. J Orthop Surg Res 2021. [Google Scholar] [Crossref]
- Background
- Materials and Methods
- Isolation and expansion of MSCs
- Immuno profiling
- Immunocytochemistry
- Population characteristics
- Lineage differentiation assays
- Cell cycle analysis
- Statistical analysis
- Results
- Phenotypic profile of isolated MSCs
- Phenotypic validation of FACS isolated MSCs
- Population characteristics
- Adipogenic, Osteogenic and Chondrogenic potential
- Cell cycle analysis
- Discussion
- Conclusion
- Key Messages
- Abbreviations
- Availability of data and materials
- Author Contributions
- Ethics Approval and Consent to Participate
- Source of Funding
- Conflict of Interest
- Acknowledgements
